Top Antibody Purification Technology Methods You Should Know?
Antibody Purification Technology is crucial for biopharmaceutical developments. According to a recent report by Grand View Research, the global antibody purification market is projected to reach $3.03 billion by 2025, with a CAGR of 12.9%. This growth underscores the increasing demand for efficient purification methods in producing therapeutic antibodies.
Dr. Sarah Thompson, a leading expert in antibody purification, emphasizes the importance of choosing the right technique: “Effective purification is key to enhancing the efficacy of therapeutic antibodies.” This statement highlights a core challenge. Selecting the optimal purification method can be complex. Many technologies exist, and each comes with distinct advantages and limitations.
Moreover, the evolving landscape of biotechnological advancements requires that professionals stay informed. While technologies like Protein A affinity chromatography dominate, new methods are emerging. Decision-makers must often weigh factors like yield, purity, and cost-effectiveness. Reflecting on these options prompts industry professionals to consider their strategies carefully. The journey through Antibody Purification Technology is as complex as it is vital.

Table of Contents
[Hide]
Top Antibody Purification Technology Methods You Should Know
Antibody purification technologies are crucial in biotechnology and pharmaceuticals. These methods ensure that antibodies are isolated efficiently and effectively. According to recent industry reports, over 70% of biopharmaceuticals utilize antibodies for therapeutic applications. The demand is expected to grow at a compound annual growth rate (CAGR) of 8% through 2025.
In ion exchange chromatography, charged molecules interact with the medium. This method requires careful optimization of pH and salt concentrations. A small miscalculation can lead to low yield or contamination. Furthermore, affinity chromatography, while popular, often depends on factors like ligand stability. Studies show that up to 30% of products may not bind effectively during purification, raising questions about scalability.
Size exclusion chromatography (SEC) is another key technique. It separates proteins based on size, offering a straightforward way to purify antibodies. However, researchers often overlook its limitations. SEC may not efficiently separate closely-sized proteins, which can complicate the final product purity. Industry analysis suggests that refining SEC parameters could elevate yields by an additional 25%. Addressing these challenges is essential for advancing antibody technology.
Understanding the Basics of Antibody Purification Techniques
Antibody purification is a crucial step in biochemical research. Understanding the basic techniques can guide researchers in choosing the right method for their needs. Common techniques include protein A affinity chromatography, ion exchange chromatography, and size exclusion chromatography. Each method has its unique advantages and limitations.
Affinity chromatography is widely used. It relies on specific interactions between antibodies and their targets, often yielding highly pure samples. However, it can be expensive and may require optimization. Ion exchange chromatography separates proteins based on charge, making it a versatile choice. Yet, it may not provide the purity needed for sensitive applications.
Researchers often overlook the importance of buffer conditions in these techniques. Variations in pH and ionic strength can significantly impact elution and binding properties. Size exclusion chromatography helps in removing aggregates and impurities but may result in some loss of target antibodies. These nuances require careful consideration during method selection and optimization. Each choice is a balance of simplicity, efficiency, and purity, demanding constant reflection and adjustment in the lab.
Top Antibody Purification Technology Methods You Should Know
| Purification Method | Principle | Applications | Advantages | Disadvantages |
|---|---|---|---|---|
| Protein A Affinity Chromatography | Uses Protein A to bind antibodies via their Fc region. | Monoclonal antibody purification. | High specificity and yield. | Costly resins; not suitable for all antibodies. |
| Ion Exchange Chromatography | Separates proteins based on charge. | Purification of various proteins. | Versatile; can achieve high purity. | Requires careful pH and salt gradient control. |
| Size Exclusion Chromatography | Separates based on size and molecular weight. | Desalting and buffer exchange. | Gentle technique; no denaturation. | Limited resolution for closely sized proteins. |
| Affinity Chromatography (Other Ligands) | Uses various ligands to capture specific molecules. | Targeted purification of proteins. | High specificity; can be tailored. | May require optimization and custom ligands. |
Plasmid and Cell Line Selection for Enhanced Antibody Production
Plasmid and cell line selection play vital roles in antibody production. Choosing the right plasmid can enhance expression levels significantly. According to a recent industry report, up to 50% of antibody yield can depend on the plasmid's design. A well-optimized plasmid contains a strong promoter and proper elements for replication and selection.
Cell line selection is equally important. Commonly used cell lines include CHO and HEK293. Each has its advantages and drawbacks. CHO cells are preferred for glycosylation patterns, while HEK293 cells often yield higher expression levels. Unfortunately, not all cell lines are equally efficient. Some may produce lower quality antibodies, leading to further purification challenges.
Tips: Consider using transient transfection methods for faster results. This can reduce time-to-market for therapeutic antibodies. Also, validate the performance of your selected cell line regularly. Continuous monitoring ensures consistency in production. Remember, not every combination will produce the anticipated results. It’s crucial to experiment and refine your approach for optimal yields.
Affinity Chromatography: Principles and Performance Metrics
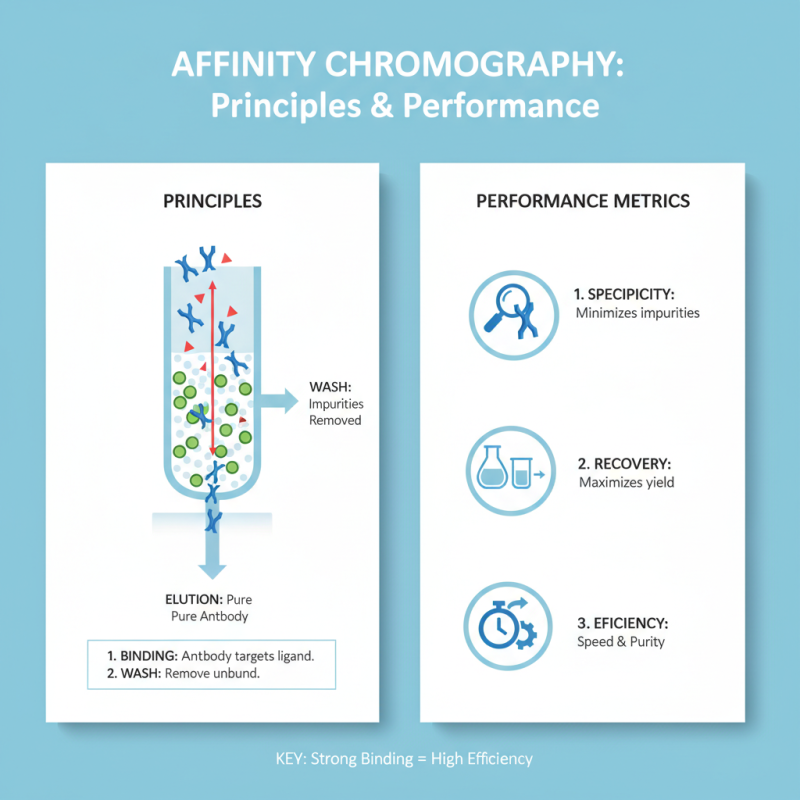
Affinity chromatography is a widely used technique for antibody purification due to its effectiveness and specificity. This method relies on the interaction between an antibody and its target antigen. The matrix is often composed of a solid support linked to an affinity ligand that captures the desired antibody. The binding affinity is crucial for purification efficiency.
When assessing performance metrics, factors like yield, purity, and binding capacity come into play. A high yield indicates that most of the antibody is recovered. Purity, often judged by the absence of contaminants, is vital for applications in research and therapeutics. Binding capacity denotes how much antibody can be captured and is influenced by the ligand's properties and resin characteristics.
**Tips:** Always run pilot tests to optimize conditions. This step can help identify the best buffer compositions and flow rates. Regularly monitor binding and elution profiles for consistent results. Be mindful of temperature fluctuations, as they can impact binding efficiency. Document all findings for future reference, ensuring reproducibility in your processes.
Alternative Techniques: Ion Exchange and Size Exclusion Chromatography
Ion exchange chromatography (IEC) and size exclusion chromatography (SEC) are vital techniques in antibody purification. According to industry reports, IEC is employed for separating proteins based on charge. This method allows for high resolution and efficiency. A study found that IEC can achieve up to 98% purity for monoclonal antibodies, a crucial factor for therapeutic applications.
SEC operates differently, focusing on the size of molecules. This method is often used as a polishing step post-IEC. It effectively separates aggregated and misfolded antibodies from correctly folded variants. Recent findings indicate that SEC provides an average recovery of around 85%, which can improve the overall yield of desired antibodies. However, reliance solely on SEC may lead to loss of valuable product, thus a careful balance is needed.
Both methods present unique challenges. IEC parameters must be optimized to avoid undesired interactions, which may lead to lower yields. SEC, while gentle, can be time-consuming. Each technique serves specific purposes but has limitations that require reflection and adjustment. Understanding these methods is essential for researchers aiming to enhance antibody purification processes.
Conclusion
Antibody Purification Technology is a crucial aspect in the production of antibodies for research and therapeutic applications. This article outlines the various methodologies involved, starting with an understanding of the foundational principles behind antibody purification techniques. Key considerations include the selection of plasmids and cell lines that enhance antibody yield, thereby improving overall production efficiency.
The discussion further delves into affinity chromatography, highlighting its principles and performance metrics, which are essential for effective purification. Additionally, alternative techniques such as ion exchange and size exclusion chromatography are evaluated, providing a comprehensive overview of the tools available for antibody purification. Finally, the article emphasizes the importance of evaluating purification yields against industry benchmarks and trends, ensuring that production processes are both efficient and scalable.
Related Posts
-
2026 Top Antibody Purification Technology Trends and Innovations?
-

How to Master Antibody Purification Technology for Optimal Results?
-

Why is Radioligand Therapy Development Essential for Targeted Cancer Treatment?
-

10 Essential Tips for Efficient Biopharmaceutical Processing
-

China Top Affinity Ligand Technology Advancements and Applications?
-
Top 10 Advances in Radioligand Therapy Development for Cancer Treatment?