10 Essential Tips for Efficient Biopharmaceutical Processing
In the ever-evolving landscape of biopharmaceutical processing, efficiency is paramount. Dr. Jane Smith, a leading expert in this field, emphasizes, "The key to success lies in optimizing every step of the process." Biopharmaceutical processing is not only about producing therapeutic drugs but also about ensuring quality and reducing costs. With the industry's complexity, adhering to efficient practices can make a significant difference.
Many companies struggle with standardization in their processes. This often leads to delays and increased production costs. The challenge lies in balancing innovation with established methods. Streamlining operations and integrating new technologies can be daunting. Achieving this balance requires careful consideration of current practices and a willingness to adapt.
Embracing the right strategies can transform challenges into opportunities. Focusing on effective practices allows biopharmaceutical companies to meet growing demand. By refining their processes, they can enhance productivity without compromising quality. In an industry defined by rapid changes, staying ahead is essential. Those who prioritize efficiency will likely lead the way.

Table of Contents
[Hide]
Understanding the Importance of Efficient Biopharmaceutical Processing
Efficient biopharmaceutical processing is vital for the industry. With global spending expected to reach over $300 billion by 2026, optimizing production can significantly enhance profit margins. Studies show that intricate processes account for over 40% of production time. Streamlining these processes is not just beneficial; it’s essential for success.
Many companies struggle with production bottlenecks. In fact, a report by the Biopharmaceutical Manufacturing Report highlights that 25% of manufacturers experience delays due to inefficient processing. This can lead to extended launch timelines and increased costs—up to 30% more than originally budgeted. Emphasizing efficient workflows can reduce these financial strains significantly.
Quality is another critical concern. A failure in a biopharmaceutical product can trigger costly recalls and severe regulatory scrutiny. An alarming statistic indicates that roughly 24% of products analyzed faced compliance issues. Implementing continuous monitoring and robust quality controls can mitigate risks and ensure product integrity. Balancing speed and quality is a challenge but essential in today’s competitive landscape.
10 Essential Tips for Efficient Biopharmaceutical Processing - Understanding the Importance of Efficient Biopharmaceutical Processing
| Tip | Description | Expected Outcome |
|---|---|---|
| 1. Optimize Cell Culture | Implement strategies to enhance cell growth and productivity. | Increased yield of biopharmaceuticals. |
| 2. Streamline Purification Processes | Utilize advanced chromatography techniques. | Higher purity with reduced processing time. |
| 3. Automate Workflow | Incorporate automation technology into processing steps. | Minimized human error and increased throughput. |
| 4. Implement Quality by Design (QbD) | Build quality into the process from the beginning. | Consistent product quality and regulatory compliance. |
| 5. Utilize Data Analytics | Leverage analytics to monitor and optimize processes. | Informed decision-making and performance improvement. |
| 6. Invest in Training | Enhance skills of personnel through continuous training. | Increased efficiency and reduced errors. |
| 7. Maintain Equipment Regularly | Schedule regular maintenance for all equipment. | Reliable operation and reduced downtime. |
| 8. Foster Cross-Department Collaboration | Encourage communication between R&D and production. | Streamlined processes and innovation. |
| 9. Optimize Supply Chain Management | Ensure timely delivery of materials and resources. | Reduced lead times and costs. |
| 10. Monitor Regulatory Changes | Stay informed about updates in regulations. | Avoid non-compliance and penalties. |
Key Stages in the Biopharmaceutical Production Process
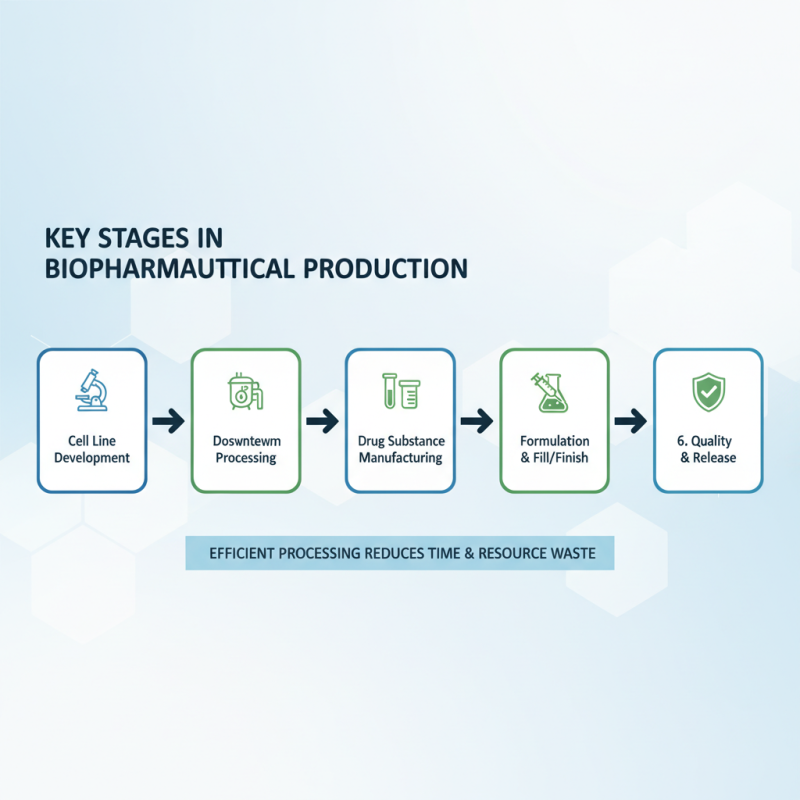
The biopharmaceutical production process involves several critical stages. Understanding these stages is vital for efficient processing. A focused approach can significantly reduce time and resource waste.
In the initial stage, cell culture is crucial. This step requires precise environment control. Maintaining optimal temperatures and pH levels is essential for cell growth. A tip for this stage is to regularly monitor the conditions, ensuring stability. Inconsistent parameters can lead to poor yields and extended timelines.
Next is the purification stage. This step separates the desired product from impurities. Each filtration needs careful execution. Implementing automated systems can enhance accuracy and reduce manual errors. Another tip is to regularly evaluate the purification methods. Adjusting timelines based on feedback can improve overall efficiency.
Finally, consider the packaging and distribution phase. This stage is often overlooked but critical. Proper packaging preserves product integrity. A reflection point here is to evaluate packaging materials continuously. Regular assessments can uncover unexpected weaknesses, helping to refine your overall process.
Best Practices for Streamlining Workflow and Reducing Downtime
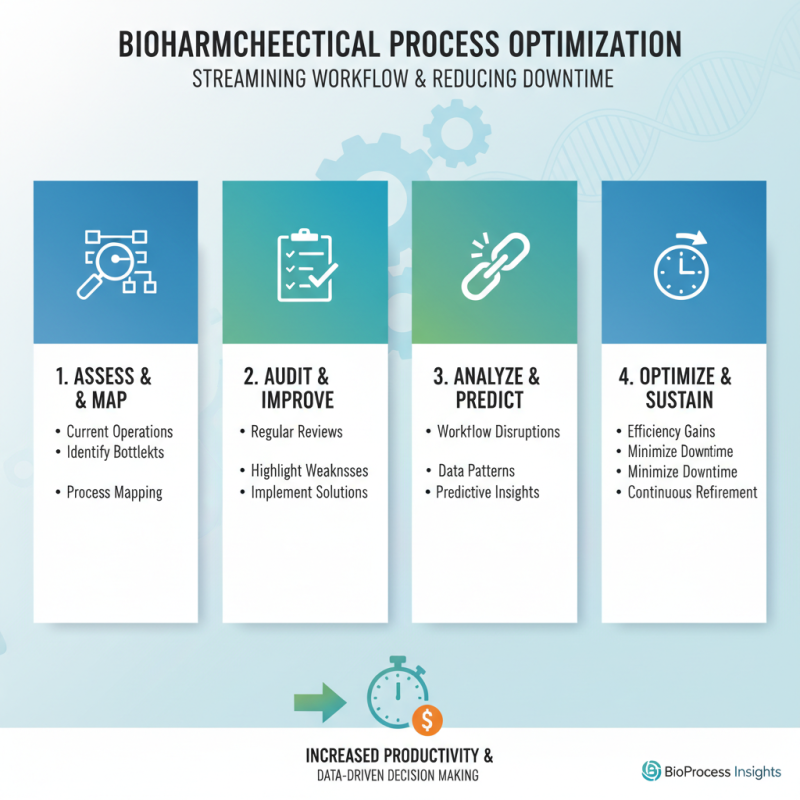
Efficient workflow in biopharmaceutical processing is critical. Streamlining these processes can significantly reduce downtime. Start by assessing current operations. Identify bottlenecks that can slow down production. Regular audits can highlight areas needing improvement. Look for patterns in workflow disruptions. Data analysis often reveals surprising insights.
Utilizing automation tools is another effective strategy. These tools can handle repetitive tasks, thus freeing up staff for more complex work. Training employees on new technologies is essential. Resistance to change can impede progress. Encourage a culture of adaptability. Celebrate small wins as teams embrace new practices.
Collaboration is vital. Cross-departmental communication can minimize misunderstandings. Weekly meetings can keep everyone aligned on goals. However, it’s crucial to listen to feedback. Employees at all levels often have valuable perspectives. Not everything will go as planned. Emphasizing continuous improvement can transform setbacks into learning experiences.
Implementing Quality Control Measures for Optimal Outcomes
Quality control is crucial in biopharmaceutical processing. It ensures the effectiveness and safety of products. Implementing robust quality control measures leads to optimal outcomes. Regular checks minimize risks in production processes. Teams must establish clear protocols for monitoring every stage.
Training personnel in these protocols is key. Knowledge gaps can lead to critical errors. Employing automated systems enhances efficiency but requires oversight. Data integrity must be maintained throughout. Any lapses in recording can compromise overall results.
It’s important to continuously assess these measures. Feedback from production lines can highlight areas for improvement. Regular audits help identify weaknesses in the process. Addressing these issues promptly can make a big difference. Quality control is not infallible; it requires constant refinement and adaptation.
10 Essential Tips for Efficient Biopharmaceutical Processing
This chart illustrates the impact of quality control measures on the efficiency of biopharmaceutical processing. Each bar represents a specific quality control practice implemented, showing the percentage improvement in processing efficiency.
Leveraging Technology for Enhanced Biopharmaceutical Processing Efficiency
In today's biopharmaceutical landscape, leveraging technology is vital for enhancing processing efficiency. Advanced automation systems can streamline workflows. These systems reduce human error and ensure consistent quality. Data management tools also play a critical role. They provide real-time insights to optimize production processes.
One key tip for biopharmaceutical companies is to invest in high-throughput screening technologies. These allow for quicker identification of potential candidates. Faster testing translates into reduced timelines for bringing therapies to market. Another effective approach is adopting modular process designs. This flexibility can significantly enhance responsiveness to demand changes.
However, challenges remain. Integrating new technologies requires careful planning and training. Staff may resist change, leading to inefficiencies. Regular assessments and feedback loops can help identify issues early. Companies must remain adaptable and ready to rethink processes to improve efficiency continually.
Related Posts
-

How to Master Antibody Purification Technology for Optimal Results?
-

China Top Affinity Ligand Technology Advancements and Applications?
-
2026 Top Antibody Purification Technology Trends and Innovations?
-

Why is Radioligand Therapy Development Essential for Targeted Cancer Treatment?
-

2026 Best Biopharmaceutical Innovation Trends to Watch Out For?