10 Best Recombinant Protein Purification Methods for You?
Recombinant protein purification is crucial in biotechnology and pharmaceutical industries. According to a 2022 market study, the global recombinant protein market is projected to reach $53 billion by 2026. This growth underlines the increasing need for effective purification methods to isolate proteins. High purity and yield are essential for practical applications in drug development and diagnostics.
Various techniques exist for recombinant protein purification. Some methods are well-established, while others are emerging. For instance, affinity chromatography remains a popular choice, enabling specific interactions with target proteins. However, challenges like protein denaturation and low recovery rates can arise. Understanding these limitations is key in selecting the right method.
Notably, the complexity of protein structures compels researchers to adopt diverse strategies. Each method has unique advantages and drawbacks. New approaches like nanomaterials in purification are gaining traction but require careful evaluation. It’s vital to balance efficiency and cost-effectiveness. The goal is to enhance protein yields while maintaining their functionality. As research advances, refined techniques will play a significant role in the purification landscape.

Table of Contents
[Hide]
Overview of Recombinant Protein Purification Techniques
Recombinant protein purification techniques play a crucial role in biotechnology. Scientists often face challenges during these processes. Understanding various methods can enhance overall efficiency.
Common techniques include affinity chromatography, ion exchange, and size exclusion chromatography. Each method has its strengths and weaknesses. Affinity chromatography is popular for its specificity but may require time-consuming steps. Conversely, ion exchange is faster but could result in lower purity levels. Size exclusion is gentle but may not separate proteins effectively based on size alone.
Researchers also explore newer methods. Emerging techniques, like magnetic bead separation and ultrafiltration, show promise. However, these innovative approaches often need extensive validation. It is essential to consider both established and novel methods carefully. The choice of purification method depends heavily on the protein's characteristics and the final application. Unexpected outcomes may lead to re-evaluating strategies and techniques used in the laboratory.
10 Best Recombinant Protein Purification Methods for You
| Method | Principle | Advantages | Limitations |
|---|---|---|---|
| Affinity Chromatography | Based on specific interactions between the protein and an affinity resin. | High specificity, suitable for purification of tagged proteins. | Requires specific tags, can be expensive. |
| Ion Exchange Chromatography | Separates proteins based on charge properties. | High resolution and capacity. | May require multiple steps to optimize conditions. |
| Gel Filtration/Size Exclusion Chromatography | Separates proteins based on size. | Gentle method, good for native proteins. | Lower resolution for closely sized proteins. |
| Affinity Precipitation | Uses specific binding agents to precipitate the target protein. | Applicable for large volumes, generally easy to perform. | Poor resolution compared to chromatography techniques. |
| HPLC (High Performance Liquid Chromatography) | High-resolution separation based on various interactions. | Very high resolution and speed. | Requires expensive equipment and consumables. |
| Ultrafiltration | Separates based on molecular weight cut-off. | Fast and efficient, scalable. | Can concentrate unwanted proteins. |
| Precipitation Methods | Involves adding reagents that cause proteins to aggregate. | Simple and cost-effective. | Low specificity, risk of co-precipitating impurities. |
| Magnetic Bead-Based Methods | Use magnetic beads coated with specific ligands. | Fast and reproducible. | Magnet accessibility can be a limitation. |
| Salt Fractionation | Uses different salt concentrations to selectively precipitate proteins. | Cost-effective and simple. | Requires careful optimization. |
| Chromatofocusing | Separates proteins by isoelectric point. | High resolution for pH-sensitive proteins. | Limited to proteins affected by pH. |
Comparative Analysis of Affinity Chromatography Methods
Affinity chromatography is a powerful tool for protein purification. This method utilizes specific interactions between a protein of interest and a ligand attached to a stationary phase. Its efficiency is heightened by the specificity of these interactions. Techniques like His-tag and GST-tag chromatography are common choices. However, the success of these methods often depends on optimal conditions. Adjusting factors such as pH, ionic strength, and temperature can lead to better yields.
Tips: Always run preliminary experiments to determine the best conditions for your target protein. A small-scale trial can save resources later.
While affinity chromatography is reliable, it still faces challenges. Non-specific binding can lead to impurities in your final product. It’s essential to optimize your wash and elution conditions. Keep in mind that prolonged elution times may cause protein degradation.
Tips: Regularly check for degradation by running SDS-PAGE analysis on your eluted fractions. This ensures the integrity of your protein throughout the purification process.
Exploring alternative methods can also be beneficial. Consider combining affinity chromatography with other techniques like size exclusion or ion exchange. This multi-step approach can enhance purity and yield. Reflect on past attempts and adjust your strategy accordingly.
Comparative Analysis of Affinity Chromatography Methods
Impact of Ion Exchange Chromatography on Purity Levels
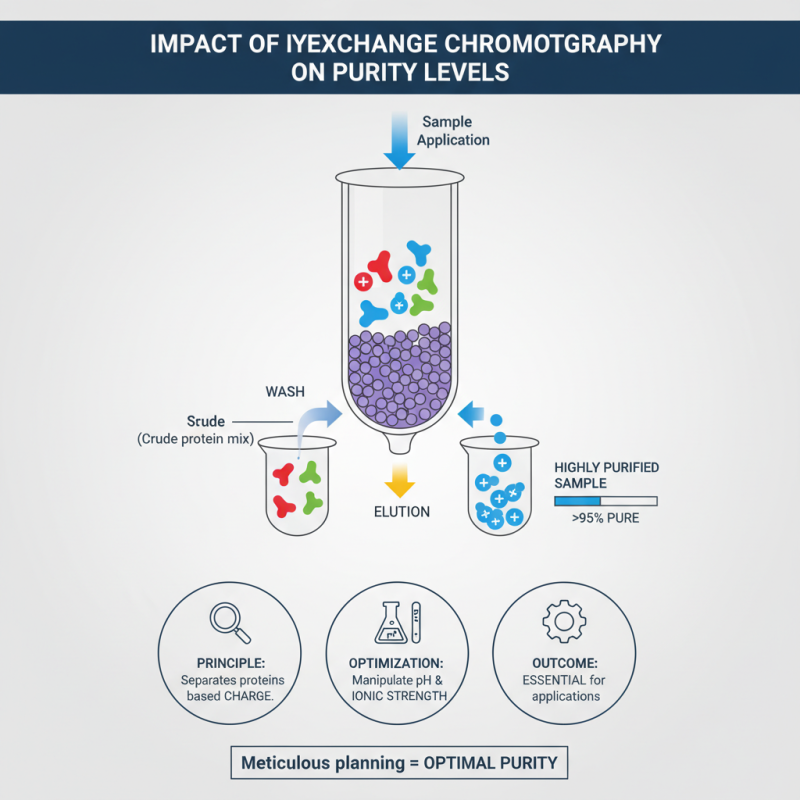
Ion exchange chromatography (IEC) is a crucial method in recombinant protein purification. It significantly impacts purity levels by separating proteins based on their charge. By manipulating pH and ionic strength, researchers can optimize binding conditions. This results in highly purified samples, essential for downstream applications. However, achieving optimal conditions requires meticulous planning and execution.
One tip for effective IEC is to pre-screen samples. This helps identify the best buffer conditions for your protein. Adjusting the pH carefully can enhance binding efficiency. Sometimes, a slight change in ionic strength can lead to considerable improvements in purity. Remember that not all proteins behave predictably under IEC. Testing multiple conditions can reveal unexpected behaviors.
It's also vital to monitor the process closely. Regular sampling allows you to track purification progress. If purity levels are not as expected, it may be necessary to revisit initial conditions. An iterative approach can help refine the purification strategy. Being flexible and open to adjustments can lead to better outcomes in achieving high purity with IEC.
Role of Size Exclusion Chromatography in Protein Separation
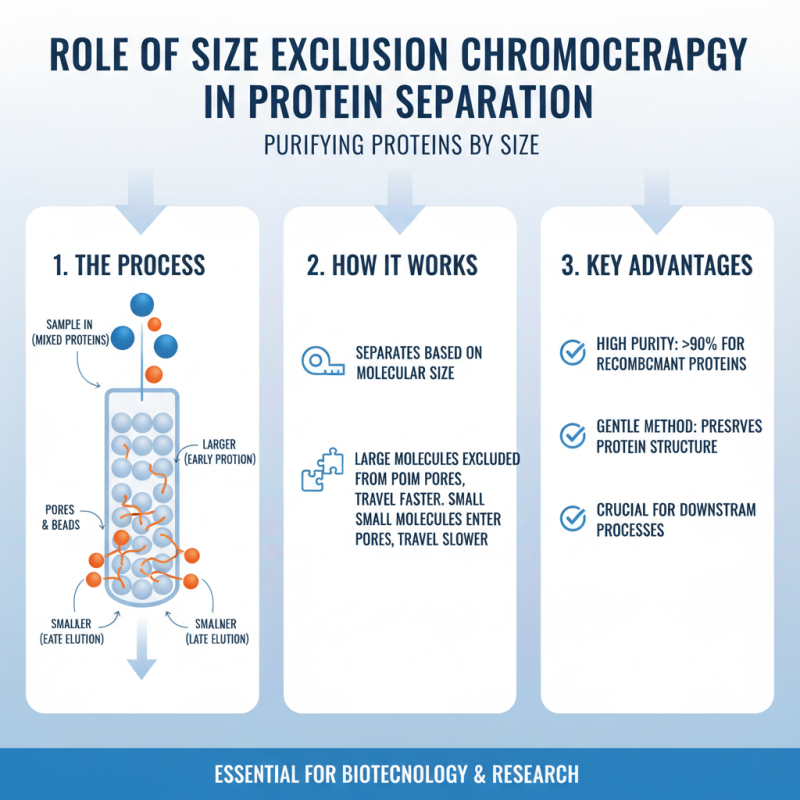
Size exclusion chromatography (SEC) is a crucial method for protein separation. This technique leverages the size of molecules to facilitate their separation. Larger proteins elute earlier compared to smaller ones. This property makes SEC an effective choice for purifying recombinant proteins. According to industry reports, SEC can achieve over 90% purity in many applications, which is vital for downstream processes.
During SEC, the protein solution passes through porous beads. Smaller molecules penetrate the beads and take longer to elute. This process helps remove contaminants such as salts and small peptides. However, the resolution may vary based on bead size and column length. Sometimes, optimizing these parameters is necessary to achieve desired results.
Despite its advantages, SEC is not without limitations. It may not effectively separate proteins of similar sizes. In such cases, combining it with other purification methods can enhance results. The need for additional methods reflects the complexity of protein purification. Researchers must continuously adapt their protocols to address these challenges effectively.
Emerging Trends in Membrane Filtration for Protein Purification
Membrane filtration is revolutionizing protein purification. This method offers efficient separation of proteins based on size and molecular weight. Researchers increasingly favor this approach due to its high throughput and scalability. Membranes can be tailored to specific needs, allowing for fine-tuning of the purification process. This flexibility reduces the time and cost associated with traditional methods.
Recent developments in membrane technology enhance performance. Novel materials are emerging that improve selectivity and reduce fouling. These advancements promise increased yields and faster processing times. However, some challenges persist. Understanding membrane behavior in various conditions is essential but not fully explored. This gap can lead to unexpected results in protein recovery.
The integration of advanced analytical techniques with membrane filtration is another trend. Real-time monitoring can help optimize the purification process. These insights are crucial for enhancing method reliability. Though progress is evident, continuous learning and adaptation are necessary. Each purification challenge offers an opportunity for improvement. Embracing this mindset is key for researchers in the field.
Related Posts
-

2026 Best Biopharmaceutical Innovation Trends to Watch Out For?
-
How to Understand Protein Binding Technology and Its Applications?
-
2026 Top Antibody Purification Technology Trends and Innovations?
-
Top 10 Advances in Radioligand Therapy Development for Cancer Treatment?
-

10 Essential Tips for Efficient Biopharmaceutical Processing
-

How to Master Antibody Purification Technology for Optimal Results?