2026 Best Biopharmaceutical Innovation Trends to Watch Out For?
The biopharmaceutical landscape is evolving rapidly. With the rise of innovative technologies, 2026 promises groundbreaking advancements in Biopharmaceutical Innovation. Experts in the field highlight various trends to watch, driven by scientific breakthroughs and patient needs. These developments aim to enhance treatment efficacy and improve patient outcomes.
New genetic therapies and advanced drug delivery systems will reshape how we approach diseases. Increased collaboration among biotech companies and research institutions is crucial. This collaboration fosters a culture of innovation that challenges traditional methodologies. However, these advancements also raise questions about accessibility and ethical implications. Can we ensure that all patients benefit from these breakthroughs?
Understanding the key trends in Biopharmaceutical Innovation can guide stakeholders. They must navigate the complexities of changing regulations and market demands. As we look ahead, the focus remains on merging science with patient-centric solutions. The journey will not be without hurdles, but the potential rewards are significant and transformative.

Table of Contents
[Hide]
Emerging Biopharmaceutical Technologies Transforming Healthcare Landscape
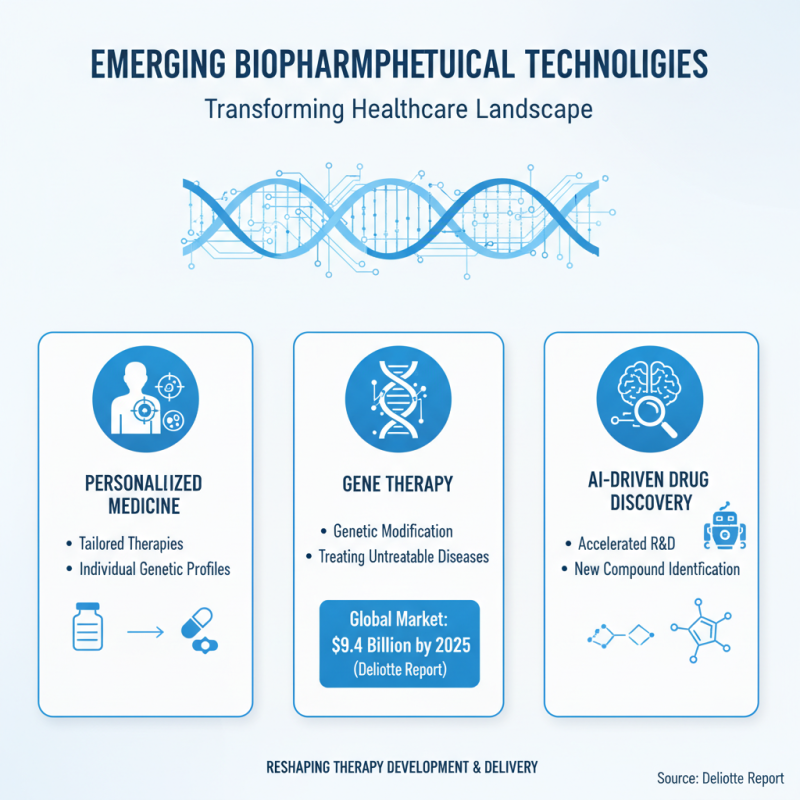
The biopharmaceutical landscape is experiencing remarkable transformations driven by emerging technologies. Innovations in personalized medicine, gene therapy, and AI-driven drug discovery are reshaping how therapies are developed and delivered. A recent report by Deloitte highlights that the global market for gene therapies is projected to reach $9.4 billion by 2025. This trend shows the potential of genetic modifications in addressing previously untreatable diseases.
AI and machine learning are accelerating drug discovery processes. Biopharmaceutical companies are leveraging data analytics to identify potential drug candidates more efficiently. According to a study published in Nature Reviews Drug Discovery, AI can cut down drug development times by about 30%. This is significant, especially when considering the traditional average of 10-15 years for bringing a new drug to market.
However, these advancements come with challenges. Ethical considerations surrounding gene editing and data privacy remain contentious. The rapid pace of innovation sometimes outstrips regulatory frameworks, leaving gaps in oversight. Industry leaders must navigate these complexities to ensure responsible development and implementation of new therapies. As biopharmaceutical technologies evolve, the need for collaboration among stakeholders becomes imperative for fostering a safe and effective healthcare environment.
Key Innovations in mRNA Therapy Revolutionizing Disease Treatment

mRNA therapy is transforming the landscape of disease treatment. This innovative approach harnesses messenger RNA to instruct cells to produce proteins that can combat illnesses. Early successes in cancer and infectious diseases reveal its potential. Vaccine development during the pandemic showcased rapid responses using mRNA technology. These advancements suggest the possibility of personalized medicine tailored for individual patients.
Despite its promise, challenges remain. Researchers must navigate complexities such as stability and delivery mechanisms. Not all patients may respond effectively to mRNA treatments. Additionally, public perception and trust in these novel therapies require careful management. Continuous education is vital to bridge the gap between scientific innovation and patient acceptance. As the field progresses, embracing reflection on ongoing challenges is crucial for comprehensive advancements in mRNA therapy.
Advancements in Gene Editing Techniques Shaping Future Therapies
Gene editing techniques are revolutionizing the field of biopharmaceuticals. The ability to precisely modify genetic material offers incredible potential for treating diseases. Techniques such as CRISPR have become more accessible, allowing researchers to explore innovative therapies. This progress is not without challenges. Ethical concerns and unintended consequences must be carefully addressed.
One promising area is gene therapy for rare genetic disorders. Patients often face limited treatment options. Gene editing can target specific mutations, offering hope for a cure. However, the complexity of human genetics means that not all edits will be successful. Researchers are learning from failures and adjusting their approaches.
The advent of gene editing also brings regulatory scrutiny. Ensuring safety and efficacy is paramount. Regulatory bodies must adapt to these rapid advancements. A balance between innovation and safety is crucial. The future of biopharmaceuticals may depend on addressing these hurdles effectively.
The Role of Artificial Intelligence in Drug Discovery and Development
Artificial Intelligence (AI) is changing the landscape of drug discovery and development. Researchers are leveraging AI to accelerate the identification of potential drug candidates. Algorithms can analyze vast data sets, identifying patterns and predicting outcomes. This efficiency can shorten timelines significantly, leading to faster clinical trials.
However, the integration of AI is not without challenges. Data quality varies greatly, and biases can lead to flawed conclusions. Ensuring the reliability of AI-based predictions is essential for effective drug development. Moreover, the human element remains vital. A skilled team is needed to interpret AI results. Analysts must understand both the science and the implications of AI findings.
Despite these hurdles, the potential for AI in biopharmaceutical innovation is enormous. It can streamline processes and enhance precision. As the field evolves, companies must remain vigilant. Balancing AI's capabilities with ethical considerations and human expertise is crucial. The future of drug discovery may heavily rely on this technology, but careful management is required.
Trends in Personalized Medicine: Tailoring Treatments for Individual Patients
Personalized medicine is transforming healthcare by focusing on individual patient needs. Traditional treatments often use a one-size-fits-all approach. This can lead to inefficacies or side effects for many patients. Personalized medicine aims to tailor therapies based on genetic, environmental, and lifestyle factors. The goal is to identify which treatments will work best for each person.
Recent advancements in genomic sequencing have significantly influenced this field. Doctors can now analyze a patient's genetic makeup to determine the most effective medications. This also helps in predicting potential adverse reactions. However, implementing personalized treatments presents challenges. Not all healthcare providers have access to the necessary technology. Additionally, the costs associated with advanced testing and tailored therapies can be high.
As the biopharmaceutical landscape evolves, patient education becomes crucial. Individuals must understand their genetic profiles and treatment options. They should engage actively in their healthcare decisions. This collaboration between patients and providers could enhance treatment outcomes. Investing in research and development is essential. There is still much to learn about the complexities of the human genome. Continuous efforts are required to improve personalized medicine further.
2026 Best Biopharmaceutical Innovation Trends to Watch Out For
| Trend | Description | Impact | Forecasted Growth |
|---|---|---|---|
| Genomic Medicine | Utilizes genetic information to personalize treatment plans. | Increased effectiveness of therapies. | 15% CAGR by 2026 |
| Gene Editing | Technologies like CRISPR to alter DNA sequences. | Potential to cure genetic diseases. | 20% CAGR by 2026 |
| Artificial Intelligence | AI-driven platforms for drug discovery and patient management. | Accelerated R&D time and cost reduction. | 18% CAGR by 2026 |
| Wearable Technology | Devices to track health biometrics in real-time. | Improved patient monitoring and compliance. | 12% CAGR by 2026 |
| Telemedicine | Remote consultations through digital communication. | Expanded access to healthcare services. | 25% CAGR by 2026 |