2026 Top Antibody Purification Technology Trends and Innovations?
As we approach 2026, significant advancements in Antibody Purification Technology are set to reshape the biotechnology landscape. According to recent market reports, the global antibody purification market is projected to reach over $1.5 billion by 2026, driven by an increasing demand for monoclonal antibodies in therapeutics and diagnostics. Innovative techniques, such as affinity chromatography and membrane technology, enhance the efficiency and specificity of antibody purification processes.
The integration of AI tools in production workflows presents new opportunities. For instance, AI-enabled systems can optimize purification protocols and automate quality controls, ensuring a higher yield and purity rate. However, the rapid adoption of technology also poses challenges. Many organizations face hurdles in implementing these advanced systems effectively, raising concerns about training and cost management.
Rethinking purification strategies is essential for maintaining competitiveness. While high-throughput methods show great promise, they can also lead to increased complexity in workflows. Therefore, a balanced approach towards innovation is necessary. Continuous evaluation of technological capabilities will help in addressing inefficiencies and ultimately contribute to creating effective and sustainable solutions in antibody purification processes.
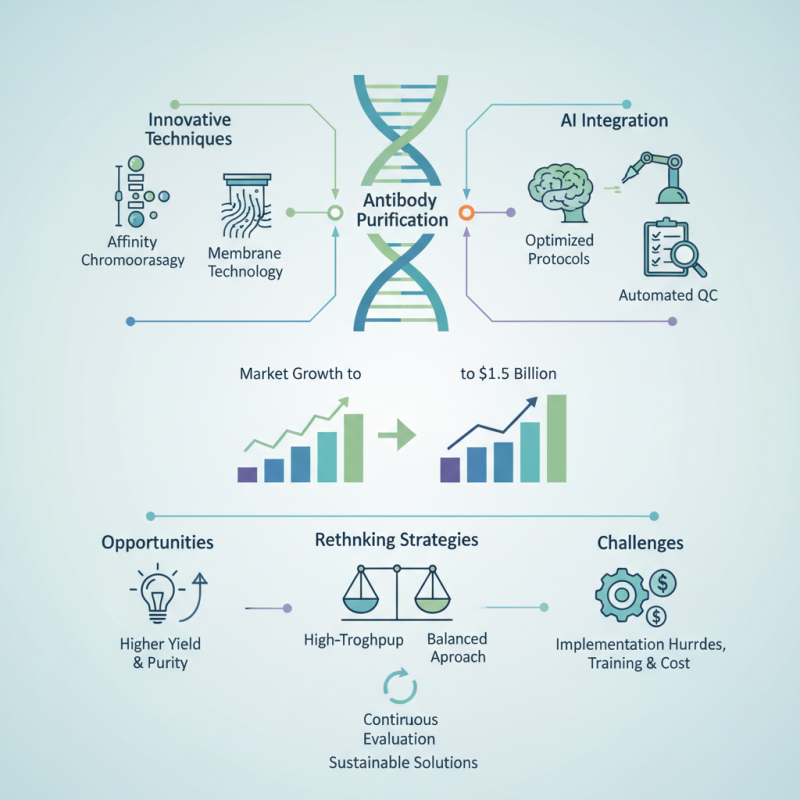
Table of Contents
[Hide]
Emerging Techniques in Antibody Purification for 2026
The landscape of antibody purification is evolving rapidly. Emerging techniques are setting the stage for advancements in 2026. One promising method is the integration of microfluidics. This technique allows for precise control over conditions. It can enhance yield and purity, but it requires careful calibration.
Another noteworthy trend is the use of nanomaterials. They can be tailored to selectively bind antibodies. This specificity could lead to better purification outcomes. Yet, there are challenges in ensuring consistency. Variability in nanomaterial production may affect the results.
Artificial intelligence is making its mark too. AI can optimize protocols and predict outcomes. It holds great potential, yet reliance on algorithms might overlook crucial human intuition. Balancing automation and personal expertise will be key. These innovations present exciting possibilities, but reflection on their limitations is essential as we move forward.
Advancements in Affinity Chromatography Methods
Advancements in affinity chromatography have transformed antibody purification. This method relies on the specific interaction between antibodies and their targets. Recently, innovative materials have enhanced binding capacities and reduced processing times. New resin technologies enable faster flow rates, which can lead to higher yields.
Next-generation affinity ligands are improving selectivity. These ligands can target specific epitopes more effectively than traditional methods. They are often designed with flexible linkers that adapt to different protein structures. Despite these advancements, challenges remain in scalability and cost.
Researchers are exploring alternative approaches as well. One emerging trend is the integration of microfluidics with affinity chromatography. This combination could lead to more controlled environments for purification. However, implementing these technologies universally is not yet straightforward. The industry still needs to address issues like reproducibility and material costs.
Innovations in Monoclonal Antibody Isolation Processes
The field of monoclonal antibody isolation is evolving rapidly. Innovations are centered around improving efficiency and purity. Traditional methods often require multiple steps, leading to longer processing times. New techniques aim to streamline these processes. For example, recent advancements utilize improved chromatography methods. These changes can lead to better yield and quality.
Another area of innovation is in the use of automated systems. Automation reduces human error and enhances reproducibility. However, it requires significant initial investment. Smaller labs may find this cost prohibitive. There’s also a need to balance automation with flexibility. High-throughput systems can sometimes overlook nuances in specific samples.
Furthermore, emerging technologies such as capillary electrophoresis are gaining attention. They promise rapid separation and analysis. Yet, these methods are still in early stages and may require further validation. Researchers must critically assess their effectiveness in practical applications. As the industry progresses, collaboration between scientists and engineers will be crucial for successful integration.
Trends in Automation and High-Throughput Purification Systems

As we move towards 2026, the field of antibody purification is witnessing significant advancements driven by automation and high-throughput systems. Recent reports indicate that the global antibody purification market is projected to grow at a CAGR of 14.5%. This rise is largely due to the increasing demand for therapeutic antibodies.
Automation is transforming purification processes. Automated systems can significantly reduce human error and increase efficiency. They enable researchers to process multiple samples simultaneously. This capability allows labs to accelerate their workflows, particularly in large-scale production environments. High-throughput systems have demonstrated efficiencies, with some reporting time savings of up to 75% in purification cycles.
Tips: Ensure that your lab is equipped with the latest automation technology. Regular training on these systems is crucial. Moreover, pay close attention to the integration of data management and analysis tools to optimize your purification processes.
Despite these innovations, challenges remain. Some labs still rely on manual processes, which can slow productivity. Additionally, not all automated systems guarantee consistency in output quality. It's vital to assess the reliability of automation solutions thoroughly. Emphasizing quality control will help mitigate risks associated with high-throughput systems.
Sustainable Practices in Antibody Purification Technology
Sustainable practices in antibody purification technology are gaining attention. Many researchers are focusing on reducing waste and energy consumption. Traditional methods often produce large amounts of hazardous byproducts. New approaches attempt to minimize this impact, yet challenges remain.
Using biobased materials is one promising avenue. These materials may reduce reliance on petrochemicals. However, sourcing and processing can still be harmful. Alternative solvents are being explored, but some have not been fully validated in all contexts. There's a need for more comprehensive studies on their overall environmental footprint.
Automation in purification processes can also enhance sustainability. Automated systems may reduce human error and improve efficiency. Nonetheless, the initial investment may be a deterrent for smaller labs. Continuous reflection on these practices is essential. Researchers need to balance innovation with environmental responsibility.

